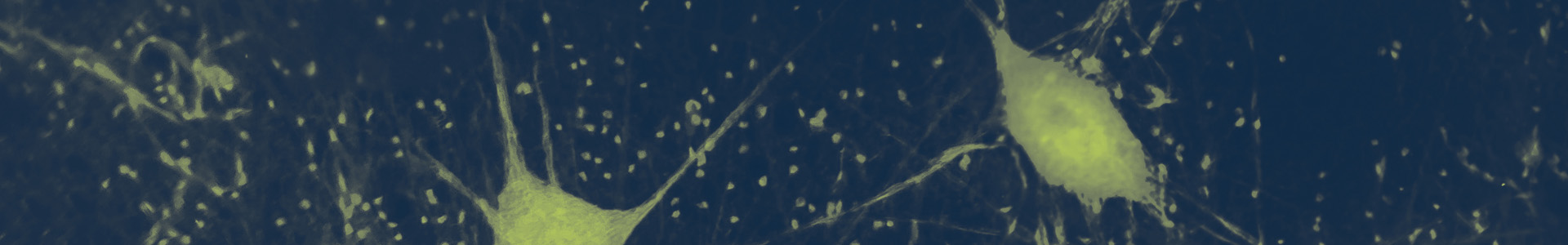
Amyotrophic lateral sclerosis (ALS) is a relentlessly progressive and fatal neurodegenerative disorder caused by motor neuron death in the brain and spinal cord. Motor neuron loss in ALS leads to deteriorating muscle function, the inability to move and speak, respiratory paralysis, and, eventually, death.1,2
“When you’re diagnosed with ALS, you’re no longer on the human clock, you’re on the ALS clock. Every passing day is a lifetime to an ALS patient, and ALS patients don’t have time to wait.”